store at low temperature
Powder: -20°C for 3 years | In solvent: -80°C for 1 year

4-Hydroxyestrone, an endogenous estrogen metabolite, can strongly protect neuronal cells against oxidative damage.
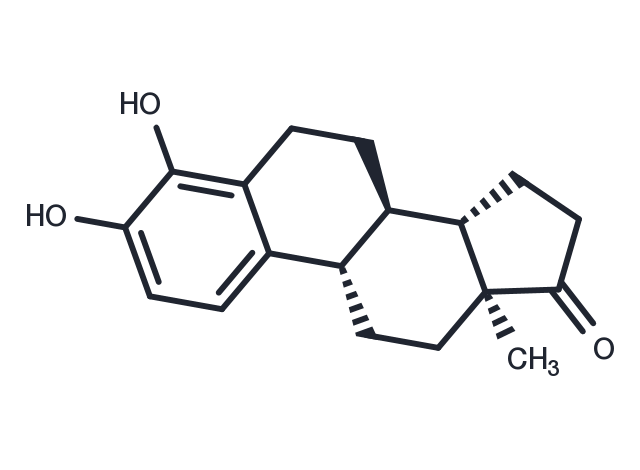
| Description | 4-Hydroxyestrone, an endogenous estrogen metabolite, can strongly protect neuronal cells against oxidative damage. |
| In vitro | Using immortalized mouse hippocampal neuronal cells as an in vitro model, 4-Hydroxyestrone, an estrone metabolite with little estrogenic activity, is found to have the strongest neuroprotective effect against oxidative neurotoxicity among 25 endogenous estrogen metabolites tested, and its protective effect is stronger than 17β-estradiol. Similarly, 4-Hydroxyestrone also exerts a stronger protective effect than 17β-estradiol against kanic acid-induced hippocampal oxidative damage in rats[1]. |
| In vivo | 4-Hydroxyestrone also exerts a stronger protective effect than 17β-estradiol against kanic acid-induced hippocampal oxidative damage in rats. Neuroprotection by 4-hydroxyestrone involves increased cytoplasmic translocation of p53 resulting from SIRT1-mediated deacetylation of p53. Analysis of brain microsomal enzymes shows that estrogen 4-hydroxylation is the main metabolic pathway in the central nervous system[1]. |
| Molecular Weight | 286.37 |
| Formula | C18H22O3 |
| CAS No. | 3131-23-5 |
store at low temperature
Powder: -20°C for 3 years | In solvent: -80°C for 1 year
You can also refer to dose conversion for different animals. More
bottom
Please see Inhibitor Handling Instructions for more frequently ask questions. Topics include: how to prepare stock solutions, how to store products, and cautions on cell-based assays & animal experiments, etc.
4-Hydroxyestrone 3131-23-5 Endocrinology/Hormones Metabolism Estrogen Receptor/ERR Endogenous Metabolite Inhibitor neuroprotective Estrone p53 SIRT1 metabolite inhibit estrogenic oxidative neurotoxicity 4-OHE1 4 Hydroxyestrone 4Hydroxyestrone deacetylation inhibitor
