Powder: -20°C for 3 years | In solvent: -80°C for 1 year


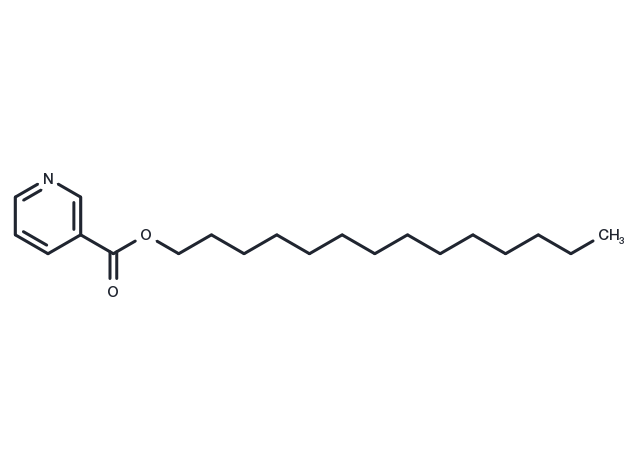
Myristyl nicotinate (Tetradecyl nicotinate) is an ester prodrug being developed for delivery of nicotinic acid (NIC) into the skin for prevention of actinic keratosis and its progression to skin cancer.
| Pack Size | Availability | Price/USD | Quantity |
|---|---|---|---|
| 100 mg | In stock | $ 46.00 | |
| 1 mL * 10 mM (in DMSO) | In stock | $ 50.00 |
| Description | Myristyl nicotinate (Tetradecyl nicotinate) is an ester prodrug being developed for delivery of nicotinic acid (NIC) into the skin for prevention of actinic keratosis and its progression to skin cancer. |
| In vitro | Myristyl nicotinate is an ester prodrug under development for delivery of nicotinic acid to skin for treatment and prevention of conditions that involve skin barrier impairment such as chronic photodamage and atopic dermatitis or for mitigating skin barrier impairment that results from therapy such as retinoids or steroids[1].?The formulation stability of Myristyl nicotinate is crucial because even small amounts of free nicotinic acid cause skin flushing, an effect that is not harmful but would severely limit tolerability[1]. |
| In vivo | Retinoic acid therapy resulted in stratum corneum thinning of approximately 25% (P = 0.006 versus baseline) that was ameliorated by Myristyl nicotinate use (P < 0.005). Therapy resulted in an increased rate of transepidermal water loss (TEWL) of approximately 45% (P = 0.001 versus baseline) and use of Myristyl nicotinate protected against the increase in TEWL with the strongest protection provided by prior use of Myristyl nicotinate (P = 0.056 versus placebo). Myristyl nicotinate use reduced the incidence of side-effects of the therapy and again prior use provided the greatest reduction of side-effects. Subjects showed statistically significant clinical improvement (P < 0.05 versus baseline) during the study. Myristyl nicotinate use did not interfere with any clinical improvement parameters and improved effects on temple laxity (P = 0.01 versus placebo). Analysis of changes in epidermal thickness, Ki67-positive cells and intensity of loricrin staining demonstrated that Myristyl nicotinate either improved or did not interfere with retinoic acid efficacy. These results show that prior and concurrent use of Myristyl nicotinate can mitigate barrier impairment and improve the tolerability of retinoic acid therapy for facial photodamage without interfering with efficacy[1]. |
| Synonyms | Tetradecyl nicotinate |
| Molecular Weight | 319.48 |
| Formula | C20H33NO2 |
| CAS No. | 273203-62-6 |
Powder: -20°C for 3 years | In solvent: -80°C for 1 year
DMSO: 15 mg/mL (46.95 mM), Sonication is recommended.
You can also refer to dose conversion for different animals. More
bottom
Please see Inhibitor Handling Instructions for more frequently ask questions. Topics include: how to prepare stock solutions, how to store products, and cautions on cell-based assays & animal experiments, etc.
Myristyl nicotinate 273203-62-6 Endocrinology/Hormones GPCR/G Protein GPR Nicotinic acid ester inhibit Tetradecyl nicotinate delivery impairment Inhibitor derivative lipophilic skin barrier prodrug inhibitor
