Powder: -20°C for 3 years | In solvent: -80°C for 1 year


Phenothiazine (ENT 38) is a class of agents exhibiting antiemetic, antipsychotic, antihistaminic, and anticholinergic activities. Phenothiazines antagonize the dopamine D2-receptor in the chemoreceptor trigger zone (CTZ) of the brain, potentially preventing chemotherapy-induced emesis.
| Pack Size | Availability | Price/USD | Quantity |
|---|---|---|---|
| 500 mg | In stock | $ 42.00 | |
| 1 mL * 10 mM (in DMSO) | In stock | $ 46.00 |
| Description | Phenothiazine (ENT 38) is a class of agents exhibiting antiemetic, antipsychotic, antihistaminic, and anticholinergic activities. Phenothiazines antagonize the dopamine D2-receptor in the chemoreceptor trigger zone (CTZ) of the brain, potentially preventing chemotherapy-induced emesis. |
| In vitro | Phenothiazines mostly substitutes at position 10 with the dialkylaminoalkyl groups and additionally at position 2 with small groups exhibit valuable activities such as neuroleptic, antiemetic, antihistaminic, antipuritic, analgesic and antihelmintic. 2-trifluoromethyl-10-(4-aminobutyl)phenothiazine inhibits S. cerevisiae strains and T. mentagrophites with MIC of 0.4 μg/mL and 1.5 μg/mL, respectively. 10-carbamoylalkylphenothiazines shows significant activities against Gram-positive Bacillus subtilis with MIC's in the range of 7.8 μg/mL–30 μg/mL. The tetracyclic phenothiazines (modified with the naphthoquinone ring) shows significant actibacterial activity against S. aureus with the MIC50 of 12.5 μg/mL. Phenothiazines with the butylene linker are more effective than with the propylene linker, the 2-chloro-10-chloroethylureidobutyl derivative giving GI50 of 1.4 μM and 1.6 μM against 4 leukemia cell lines and 7 colon cancer cell lines. 10-Amino(hydroxy)propylphenothiazines (5 μM) induces a marked G2/M phase of cell-cycle arrest followed by cell death in human transformed WI38VA cells after 2-day incubation. [1] Phenothiazine drugs undergo extensive metabolism in the body before being excreted, mainly ring hydroxylation, ring sulphoxidation, N-demethylation, N-oxidation, sulphate and glucuronide conjugation. Phenothiazines have considerably lower binding affinities to α2-adrenoceptors than to dopamine D2 receptors and al-adrenoceptors. [2] Phenothiazines have significant in vitro activity against susceptible, polydrug- and multidrug-resistant strains of M. tuberculosis, as well as enhancing the activity of some agents employed for first-line treatment. [3] |
| Synonyms | ENT 38 |
| Molecular Weight | 199.27 |
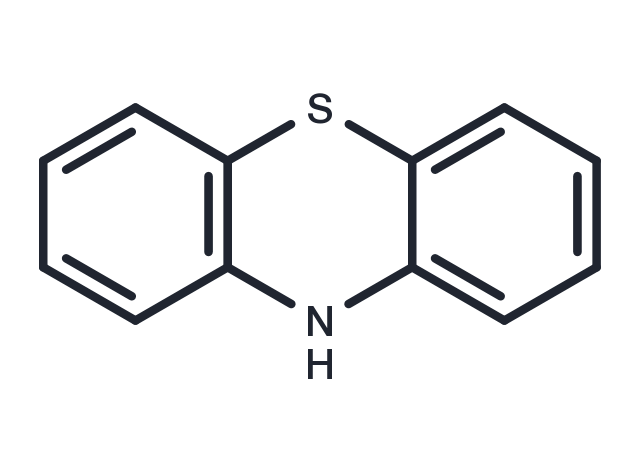
| Formula | C12H9NS |
| CAS No. | 92-84-2 |
Powder: -20°C for 3 years | In solvent: -80°C for 1 year
DMSO: 38 mg/mL (190.7 mM)
Ethanol: < 1 mg/mL (insoluble or slightly soluble)
H2O: < 1 mg/mL (insoluble or slightly soluble)
You can also refer to dose conversion for different animals. More
bottom
Please see Inhibitor Handling Instructions for more frequently ask questions. Topics include: how to prepare stock solutions, how to store products, and cautions on cell-based assays & animal experiments, etc.
Phenothiazine 92-84-2 GPCR/G Protein Microbiology/Virology Neuroscience Dopamine Receptor Antibacterial Antibiotic Antifungal inhibit antibacterial ENT 38 neurological Inhibitor Bacterial ENT-38 fungicidal Fungal ENT38 insecticidal anthelmintic inhibitor
