Powder: -20°C for 3 years | In solvent: -80°C for 1 year


Pradefovir (Remofovir) is a reverse transcriptase inhibitor potentially for treatment of chronic HBV infection. Pradefovir is also a liver-targeted prodrug of adefovir. After metabolic activation, Pradefovir was converted to PMEA (9-(2-phosphonylmethoxyethyl)adenine) in human liver microsomes with a K(m) of 60 microM, a maximum rate of metabolism of 228 pmol/min/mg protein, and an intrinsic clearance of about 359 ml/min.
| Pack Size | Availability | Price/USD | Quantity |
|---|---|---|---|
| 25 mg | In stock | $ 1,300.00 | |
| 50 mg | In stock | $ 1,690.00 |
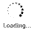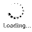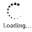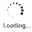
| Description | Pradefovir (Remofovir) is a reverse transcriptase inhibitor potentially for treatment of chronic HBV infection. Pradefovir is also a liver-targeted prodrug of adefovir. After metabolic activation, Pradefovir was converted to PMEA (9-(2-phosphonylmethoxyethyl)adenine) in human liver microsomes with a K(m) of 60 microM, a maximum rate of metabolism of 228 pmol/min/mg protein, and an intrinsic clearance of about 359 ml/min. |
| In vivo | Pradefovir, a prodrug of PMEA, is under phase 2 clinical trial in China to evaluate its pharmacokinetic and pharmacodynamics after multiple-dose study, with adefovir dipivoxil and tenofovir disoproxil fumarate as positive control. A rapid and sensitive liquid chromatography-tandem mass spectrometry (LC-MS/MS) method was developed and validated for the quantification of pradefovir, PMEA and tenofovir in HBV patient serum. Serum samples were pretreated via simple protein precipitation with methanol and entecavir was used as internal standard. Chromatographic separation was carried out on a Synergi(?) fusion-RP column (150mm×4.6mm) by gradient elution with methanol and 0.1% formic acid in water (v/v) at a flow rate of 1mL/min. The analytes were detected in multiple reaction monitoring mode with positive ion electrospray ionization at m/z 424.1/151.0, 274.1/162.2, 288.1/176.1, and 278.1/152.2for pradefovir, PMEA, tenofovir and IS, respectively. The assays were validated according to current bioanalytical guidelines including specificity, linearity (2.0-500ng/mL for pradefovir and PMEA, 4.0-1000ng/mL for tenofovir), accuracy and precision, extraction recovery, matrix effect and stability. The validated method has been successfully applied to the pharmacokinetic study of pradefovir, adefovir dipivoxil and tenofovir disoproxil fumarate in a set of HBV patients[1]. |
| Synonyms | ICN-2001-3, MB-6866, ICN-20013, MB-06866, Remofovir |
| Molecular Weight | 423.79 |
| Formula | C17H19ClN5O4P |
| CAS No. | 625095-60-5 |
Powder: -20°C for 3 years | In solvent: -80°C for 1 year
DMSO: Soluble
You can also refer to dose conversion for different animals. More
bottom
Please see Inhibitor Handling Instructions for more frequently ask questions. Topics include: how to prepare stock solutions, how to store products, and cautions on cell-based assays & animal experiments, etc.
Pradefovir 625095-60-5 Microbiology/Virology HBV ICN 20013 MB 6866 ICN-2001-3 MB-6866 ICN20013 MB6866 MB 06866 ICN-20013 MB-06866 Remofovir MB06866 inhibitor inhibit
