Shopping Cart
- Remove All
 Your shopping cart is currently empty
Your shopping cart is currently empty

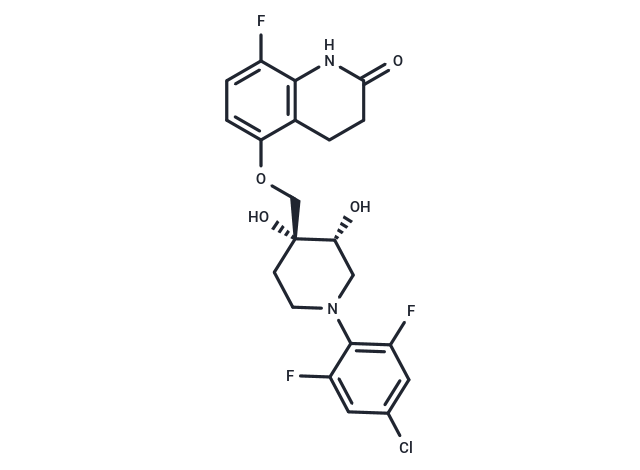
OPC-167832 is a potent and orally active dprE1 Inhibitor with an IC50 of 0.258 μM. OPC-167832 has antituberculosis activity and can be used for the research of tuberculosis caused by Mycobacterium tuberculosis[1].
| Pack Size | Price | Availability | Quantity |
|---|---|---|---|
| 25 mg | $1,170 | 10-14 weeks | |
| 50 mg | $1,530 | 10-14 weeks | |
| 100 mg | $2,440 | 10-14 weeks |
| Description | OPC-167832 is a potent and orally active dprE1 Inhibitor with an IC50 of 0.258 μM. OPC-167832 has antituberculosis activity and can be used for the research of tuberculosis caused by Mycobacterium tuberculosis[1]. OPC-167832 exhibits very low MICs against laboratory strains of M. tuberculosis H37Rv (MIC: 0.0005 μg/ml) and Kurono (MIC: 0.0005 μg/ml) and strains with monoresistance to rifampin (RIF), isoniazid (INH), ethambutol (EMB), streptomycin (STR), and pyrazinamide (PZA) (MIC: 0.00024-0.001 μg/ml). However, OPC-167832 has minimal or no activity against standard strains of nonmycobacterial aerobic and anaerobic bacteria[1].The IC90 values of OPC-167832 against intracellular M. tuberculosis strains H37Rv and Kurono are 0.0048 and 0.0027 μg/ml, respectively. OPC-167832 shows bactericidal activity against intracellular M. tuberculosis at a low concentration, and the bactericidal activity is saturated at concentrations of 0.004 μg/ml or higher[1]. OPC-167832 (oral administration; 0.625-10 mg/kg) exhibits a good pharmacokinetic characteristic. The plasma reaches peak at 0.5 h to 1.0 h (tmax) and is eliminated with a half-life (t1/2) of 1.3 h to 2.1 h OPC-167832 distribution in the lungs is approximately 2 times higher than that in plasma, and the Cmax and AUCt of OPC-167832 in plasma and the lungs shows dose dependency[1].OPC-167832 (oral administration; 0.625-10 mg/kg; 4 weeks) significantly reduces lung CFU compared to the vehicle group. The dose-dependent decrease of lung CFU is observed from 0.625 mg/kg to 2.5 mg/kg. In a M. tuberculosis Kurono-infected ICR female mice model. OPC-167832 combines with DMD, BDQ, or LVX via oral gavage exhibits significantly higher efficacies than each single agent alone[1].[1].OPC-167832 (oral gavage; 2.5 mg/kg; combination with DCMB; 12 weeks) demonstrates the most potent efficacy when compares with DC, DCB. The lung CFU count after 6 weeks of treatment is below the detection limit, and at the end of just 8 weeks of treatment, the bacteria in the lungs of all the evaluated mice had already been eradicate[1]. [1]. Norimitsu Hariguchi, et al. OPC-167832, a Novel Carbostyril Derivative with Potent Antituberculosis Activity as a DprE1 Inhibitor.Antimicrob Agents Chemother. 2020 May 21;64(6):e02020-19. |
| In vitro | OPC-167832 exhibits very low MICs against laboratory strains of M. tuberculosis H37Rv (MIC: 0.0005 μg/ml) and Kurono (MIC: 0.0005 μg/ml) and strains with monoresistance to rifampin (RIF), isoniazid (INH), ethambutol (EMB), streptomycin (STR), and pyrazinamide (PZA) (MIC: 0.00024-0.001 μg/ml). However, OPC-167832 has minimal or no activity against standard strains of nonmycobacterial aerobic and anaerobic bacteria[1].The IC90 values of OPC-167832 against intracellular M. tuberculosis strains H37Rv and Kurono are 0.0048 and 0.0027 μg/ml, respectively. OPC-167832 shows bactericidal activity against intracellular M. tuberculosis at a low concentration, and the bactericidal activity is saturated at concentrations of 0.004 μg/ml or higher[1]. |
| In vivo | OPC-167832 (oral administration; 0.625-10 mg/kg) exhibits a good pharmacokinetic characteristic. The plasma reaches peak at 0.5 h to 1.0 h (tmax) and is eliminated with a half-life (t1/2) of 1.3 h to 2.1 h OPC-167832 distribution in the lungs is approximately 2 times higher than that in plasma, and the Cmax and AUCt of OPC-167832 in plasma and the lungs shows dose dependency[1].OPC-167832 (oral administration; 0.625-10 mg/kg; 4 weeks) significantly reduces lung CFU compared to the vehicle group. The dose-dependent decrease of lung CFU is observed from 0.625 mg/kg to 2.5 mg/kg. In a M. tuberculosis Kurono-infected ICR female mice model. OPC-167832 combines with DMD, BDQ, or LVX via oral gavage exhibits significantly higher efficacies than each single agent alone[1].[1].OPC-167832 (oral gavage; 2.5 mg/kg; combination with DCMB; 12 weeks) demonstrates the most potent efficacy when compares with DC, DCB. The lung CFU count after 6 weeks of treatment is below the detection limit, and at the end of just 8 weeks of treatment, the bacteria in the lungs of all the evaluated mice had already been eradicate[1]. |
| Molecular Weight | 456.84 |
| Formula | C21H20ClF3N2O4 |
| Cas No. | 1883747-71-4 |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year | Shipping with blue ice. | |||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: 180 mg/mL (394.01 mM), Sonication is recommended. | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
DMSO
| ||||||||||||||||||||||||||||||||||||

Copyright © 2015-2025 TargetMol Chemicals Inc. All Rights Reserved.