Shopping Cart
- Remove All
 Your shopping cart is currently empty
Your shopping cart is currently empty

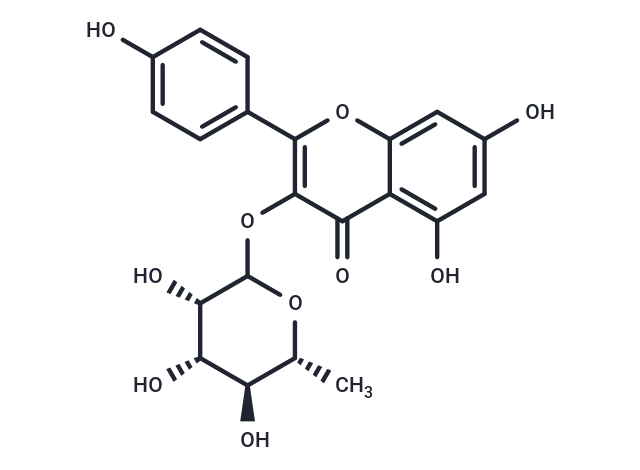
Afzelin (Kaempferol-3-O-rhamnoside) exhibits several cellular activities such as DNA-protective, antibacterial, antioxidant, anti-inflammatory, and UV-absorbing properties, potentially protecting human skin from UVB-induced damage through a combination of UV-absorbing and cellular actions. Afzelin mitigates mitochondrial damage, promotes mitochondrial biogenesis, and reduces levels of mitophagy-related proteins, parkin and PTEN-induced putative kinase 1.
| Pack Size | Price | Availability | Quantity |
|---|---|---|---|
| 1 mg | $41 | In Stock | |
| 5 mg | $127 | In Stock | |
| 10 mg | $189 | In Stock | |
| 25 mg | $315 | In Stock | |
| 50 mg | $468 | In Stock | |
| 100 mg | $686 | In Stock |
| Description | Afzelin (Kaempferol-3-O-rhamnoside) exhibits several cellular activities such as DNA-protective, antibacterial, antioxidant, anti-inflammatory, and UV-absorbing properties, potentially protecting human skin from UVB-induced damage through a combination of UV-absorbing and cellular actions. Afzelin mitigates mitochondrial damage, promotes mitochondrial biogenesis, and reduces levels of mitophagy-related proteins, parkin and PTEN-induced putative kinase 1. |
| In vitro | Therefore, in this study, we investigated the protective effects of Afzelin, one of the flavonoids, against UV irradiation in human keratinocytes and epidermal equivalent models. Spectrophotometric measurements revealed that the Afzelin extinction maxima were in the UVB and UVA range, and UV transmission below 376 nm was <10%, indicating UV-absorbing activity of Afzelin. In the phototoxicity assay using the 3T3 NRU phototoxicity test (3T3-NRU-PT), Afzelin presented a tendency to no phototoxic potential. In addition, in order to investigate cellular functions of Afzelin itself, cells were treated with Afzelin after UVB irradiation. In human keratinocyte, Afzelin effectively inhibited the UVB-mediated increase in lipid peroxidation and the formation of cyclobutane pyrimidine dimers. Afzelin also inhibited UVB-induced cell death in human keratinocytes by inhibiting intrinsic apoptotic signaling. Furthermore, Afzelin showed inhibitory effects on UVB-induced release of pro-inflammatory mediators such as interleukin-6, tumor necrosis factor-α±, and prostaglandin-E2 in human keratinocytes by interfering with the p38 kinase pathway. Using an epidermal equivalent model exposed to UVB radiation, anti-apoptotic activity of Afzelin was also confirmed together with a photoprotective effect at the morphological level[1] |
| Alias | Kaempferol-3-O-rhamnoside |
| Molecular Weight | 432.38 |
| Formula | C21H20O10 |
| Cas No. | 482-39-3 |
| Smiles | C[C@H]1OC(Oc2c(oc3cc(O)cc(O)c3c2=O)-c2ccc(O)cc2)[C@@H](O)[C@@H](O)[C@@H]1O |
| Relative Density. | 1.70 g/cm3 (Predicted) |
| Storage | keep away from direct sunlight,store at low temperature | Powder: -20°C for 3 years | In solvent: -80°C for 1 year | Shipping with blue ice. | |||||||||||||||||||||||||
| Solubility Information | Ethanol: 12 mg/mL (27.75 mM), Sonication is recommended. | |||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||
Ethanol
| ||||||||||||||||||||||||||

Copyright © 2015-2025 TargetMol Chemicals Inc. All Rights Reserved.